Persistence of Toxoplasma gondii infections: Mechanisms of action of potential drug candidates and the role of physiological heterogeneity
Background
Toxoplasma gondii is an apicomplex parasite that infects virtually all warm-blooded animals and 30% of humans worldwide. In Germany a seroconversion rate of 1% per year leads to a prevalence of 50%. This illustrates the need to reduce the zoonotic transmission of parasites in the interest of public health. T. gondii infections are typically self-limiting, but severe and fatal symptoms may occur during congenital transmission and in immunocompromised individuals. Compared to other food-borne infectious diseases, T. gondii causes one of the highest disease burden. Infection occurs, among other things, through chronically persistent tissue cysts in uncooked meat products. In contrast to acute forms of the parasite, the permanent stages, so-called bradyzoites, adopt very heterogeneous manifestations and resist all available drug treatments.
Mission
The junior research group Toxo investigates the metabolism of bradyzoites to develop evidence-based novel treatments. The work includes a systematic screen of antimicrobial compounds and a metabolomics-centered approach based on gas and liquid chromatography coupled mass spectroscopy. The identification of molecular targets of these compounds will facilitate the development and optimization of urgently needed drug candidates.
Complementary approach
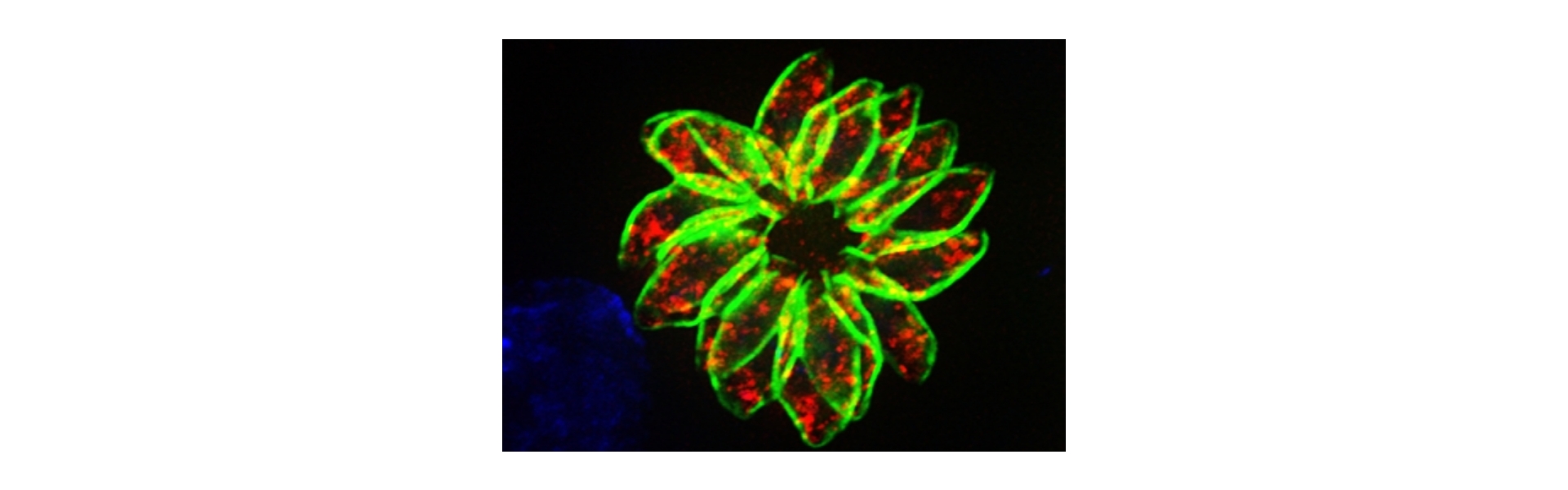
In a complementary approach, the Toxo Junior Research Group is investigating the physiological basis of persistent parasite populations in different veterinary T. gondii isolates. Tissue cysts from clonal parasite populations are phenotypically very heterogeneous. This heterogeneity could be a basis for persistence and resistance to therapy. We generate fluorescent reporter cell lines that allow us to study the response of individual cysts and parasites during the reaction to stressors such as different inhibitors. This data will then be used to calibrate a computer model that will allow us to quantify the contribution of individual subpopulations to persistence.
In summary, the Junior Research Group Toxo aims to understand persistence, which is essential for zoonotic transmission, and to derive treatment approaches.
1. Identification of the mode of action of T. gondii-cyst inhibitors
There are currently no drugs that are effective against chronic T. gondii infections. Although such latent infections are mostly asymptomatic, they can lead to acute infections and are the basis for a high infection rate, which leads to a prevalence of 50% in Germany.
There are two basic strategies to develop new drug candidates. The classical pharmacological approach is based on an initial phenotypic screen to identify potent compounds. However, the optimization of these initial hits is often difficult due to the unknown mode of action. In contrast, the reverse pharmacological approach is based on the characterization of essential proteins as potential targets. However, inhibitors synthesized for these targets are often inefficient at the organismic level.
The Toxo junior research group aims to combine the advantages of both approaches by establishing a comprehensive metabolomics platform that allows the comprehensive characterization of the parasitic metabolic response to corresponding antimicrobiota and thus the identification of target structures and modes of action. This platform consists of a UHPLC coupled high-resolution orbitrap mass spectrometer and a gas chromatography coupled mass spectrometer. Together, these instruments enable the detection of hundreds of small metabolic molecules (metabolites) that reflect corresponding enzyme activities. Such open-target analyses yield a fine-grained image of the metabolic phenotype that allows us to identify potential target pathways and target enzymes. Subsequent more targeted approaches will confirm these putative target structures by isotope labelling and reverse genetic approaches.
With this targeted metabolomics based strategy we hope to find new treatment approaches for chronic toxoplasma infections that are also transferable to other pathogens.
2. Quantification of the contribution of heterogeneity to chronic infections of T. gondii
Unlike acute forms of T. gondii, persistent stages of the parasite resist the host's immune system and all available drug treatments. These persistent stages proliferate asynchronously and with widely varying division rates and tissue cysts continue to show marked differences in packing density and size. The phenotypic heterogeneity of clonal populations of microorganisms acts as a safety mechanism for many pathogens; for T. gondii, however, its function has not been investigated. To measure the range of phenotypes at the single cell level we use fluorescent reporter parasites, which allow us to measure basic physiological parameters such as cell division activity, pH and metabolite occurrence. We use this physiological data to define subpopulations and to calibrate a computer model. This model aims to replicate the persistence and virulence phenotypes of different veterinary isolates based on these subpopulations.
Coordination
Dr. Martin Blume
Junior Research Group 2
Robert Koch-Institut
Berlin
E-Mail: blumem(at)rki.de
Tel. +49 (0)30 18754 2572
Metabolic determinants of cold tolerance of Aedes Mosquito eggs Ruth Müller, Goethe Universität Frankfurt, Institute of Occupational, Social and Environmental Medicine
Characterization of the metabolic role of Ferredoxin in Toxoplasma gondii Frank Seeber, Robert Koch-Institute, FG16
Characterization of polyunsaturated fatty acid degradation in Leishmania major Toni Aebischer, Robert Koch-Institute, FG16
Metabolomic signature of hanta virus infected cells Rainer Ulrich, Friedrich-Loeffler-Institute